Introduction
Oxygen demand measurements are used to estimate water pollution levels entering and leaving wastewater treatment and industrial facilities. Particularly close attention is paid to effluents as high oxygen demand levels indicate a danger to aquatic life. Since treatment facilities are considered point sources, they are regulated under the Clean Water Act (CWA) which in turn sets facility specific pollutant release targets as part of the National Permit Discharge Elimination System (NPDES) permitting process. NPDES permits usually specify limits related to oxygen demand based on either the Chemical Oxygen Demand (COD) or Biochemical Oxygen Demand (BOD) testing methods.
BOD Testing
BOD measures how much dissolved oxygen (DO) is consumed by microorganisms to decompose organic matter under aerobic conditions. Since BOD is a measurement based on a biological process, testing for it can take a few days. How is BOD tested? Incubate a sealed water sample at 20oC for five days, followed by measuring the difference in oxygen content before and after incubation.

Figure 1: An operator filling a BOD bottle.
COD Testing
COD measures how much DO is consumed by the chemical oxidation of organic matter under controlled conditions. Since COD is based on a chemical process testing for it will only take a few hours. COD testing typically involves digestion of the water sample in a sealed vial with potassium dichromate and sulfuric acid at 150oC for 2 hours. Vials are read in a spectrophotometer to determine results.

Figure 2: A 25 pack of CHEMetrics COD vials.
COD vs. BOD Methods for Testing Water Quality
Of the two tests, the COD method for water analysis is more commonly performed as it takes less time and is more replicable. Furthermore, treatment operators can quickly react to changes in oxygen demand and modify treatment processes appropriately. COD measurements will always be higher than BOD measurements because COD is measuring everything that can be oxidized in the sample, where BOD is measuring only what can be oxidized through biological processes. Plant engineers may establish a statistically validated COD/BOD ratio to demonstrate their ability to reliably predict a BOD value based solely on a COD measurement to their permitting authority. The COD value may be used as justification for BOD removal from a plant permit.
Conclusion
CHEMetrics offers a comprehensive line of USEPA-accepted COD vials as methods for testing water quality. K-7350S is for low range measurements (0-150 ppm) while K-7360S is for high range measurements (0-1,500 ppm). While not USEPA-accepted, K-7370S can measure up to 15,000 ppm. CHEMetrics also offers a line of mercury-free vials (K-7351S, K-7361S, and K-7371S) that are more convenient for disposal.
Introduction
Use of Film Forming Amines (FFA) or Film Forming Amine Product (FFAP) is a growing trend in the power industry for maintenance of boiler systems. Case study literature suggests that filming amines can serve as corrosion inhibitors, reduce corrosion product transport, provide smooth heat transfer surfaces, and protect equipment during shutdown or layups. FFAPs can be used with or replace conventional corrosion inhibitor treatment regimes. FFAPs are used in various kinds of steam-generating power plants including conventional fossil, combined cycle, nuclear, and biomass power plants. They are also applied to different components of the steam cycle including the boiler feedwater, generator, and condensate return lines. As water treatment chemical vendors continue to expand their product offerings, FFAP formulations are being customized to suit the individual power plant’s operational conditions.

FFAP Properties
FFAPs have a strong surface affinity for metals, forming an impermeable layer which prevents corrosive agents from contacting the surface. This binding behavior can be attributed to the chemical structures of FFAs, which are long carbon chains with an amine at one end. The amine “head” attaches to a metallic surface while the “tail” forms a protective hydrophobic film layer. Octadecylamine (ODA) is an example of the simplest FFA molecule and is constructed from a straight chain of 18 carbons. ODA was the primary FFA in use over 40 years ago when it was first introduced. More complex FFAPs were introduced to the marketplace as ODA operational challenges began to be better understood. These newer FFA compounds contain various functional groups such as primary or secondary amines which branch off from the FFA backbone. They may be referred to as polyamines or fatty amines. As use of FFAs has grown, many chemical vendors have created their own proprietary FFAPs which are formulated with other amines as well as neutralizing and emulsifier agents.
FFA Benefits
FFA technology has been shown to offer better heat transfer rates across steel tube surfaces than traditional phosphate-based corrosion inhibitor programs. Case studies in the literature also document improved bubble evaporation and the formation of smooth and homogeneous magnetite layers. FFAs will adsorb to the protective metal oxide layer and provide an extra barrier between it and the water or steam. Additionally, it has been shown that FFAPs remove loosely bound deposits within the boiler system resulting in cleaner surfaces overall. Together, these performance improvements can lead to reductions in operating costs.
FFAPs are usually touted as a greener alternative than conventional boiler treatment chemicals such as hydrazine. Since FFAs are relatively non-toxic, extensive personal protective equipment (PPE) is not required when working with these products.
FFA Pitfalls
While FFA treatment programs offer many benefits, some drawbacks are possible. A primary concern is the formation of FFA degradation products which are largely carbon dioxide and organic acids that form after extended exposure to high heat. These degradation products can lower the pH of condensed steam and increase Conductivity After Cation Exchange (CACE) unless compensated for by other water treatment methods. Another major concern is optimizing for the appropriate dosage. Overdosing offers no additional protection and runs the risk of creating micelles or “gunk balls” that can clog pipes and reduce efficiency. Supplier product and site-specific dosage rate recommendations should be followed.
Preparations Before Switching to an FFA Program
It is important to establish baseline readings of key steam cycle performance indices before changing the treatment regimen. Without these reference points, plant personnel will not have the ability to validate quantitatively whether the addition of an FFA/FFAP has provided any benefit. Some of these key parameters include total iron and/or copper, feedwater pH, oxygen, and CACE. Also, inspecting interior surfaces for loose deposits and sludge on high pressure (HP) evaporators or conventional boiler waterwalls is recommended before starting a course of FFA/FFAP. If the deposits are not removed, under deposit corrosion may occur.

FFA Dosing
Once the decision has been made to begin using FFA, the primary focus shifts to establishing the optimal dosage. Usually FFAs are fed undiluted into feedwater through an automatic pump. When introducing FFA for the first time, it is recommended to start at a low dosage and gradually increase to the target dosage, which typically takes a minimum of 3 to 4 weeks. FFA/FFAP exert a moderate cleaning effect that can lift particulate deposits of iron and/or copper oxides within the system. These deposits should be monitored to assess the FFA cleaning period duration. The effect of the FFA dosage on the iron and copper must be compared to corrosion product control guidance published in the applicable boiler technical manual. Determining dosage levels should occur in conjunction with the chemical supplier to ensure that the product is compatible with the conditions and materials within the system. It is commonly found that no FFA will be detected in the condensate phase initially as the FFA will bond to the metal surfaces and not be freely circulating.

FFA Sampling
Testing for FFA offers unique challenges as well. Since FFAs adhere to metallic, glass, and plastic surfaces, surface contact must be kept to a minimum when obtaining water samples. This means limiting sample transfer to a single container if possible; samples should be cooled to prevent flashing; sample lines should be flushed thoroughly before sampling; and sampling points should be representative of the system.
FFA Analysis
The two most common test methods for FFAs are methyl orange extraction and xanthene dye reaction. The methyl orange extraction method makes use of an organic solvent to extract the FFA to form a colored complex with the methyl orange. The xanthene reaction method utilizes a class of dyes (represented by Rose Bengal) that form a water-soluble colored complex with FFA. The FFA concentration is proportional to color intensity in both test methods. Commercially available test kits often express results in terms of common FFAs like ODA. For best accuracy, it is recommended that analysts determine a correction factor to express results in terms of the product specific FFA. Test kits that tolerate interference from ammonia or short chain amines and offer a wide measurement range to avoid dilution steps are preferable. Dilution can lead to a loss of FFA during sample transfer from one vessel to another which may affect test results.
CHEMetrics offers a Methyl Orange Filming Amine visual test kit, Cat. No K-1001, that can be used to verify residual FFA in a system in 3 minutes. This kit utilizes a unique extraction technique which eliminates several steps required in other procedures and provides increased sensitivity while limiting the extraction to a single extraction tube preventing FFA loss. If necessary, the measurement range can be extended by performing a dilution in the extraction tube provided in the test kit. Test results are expressed in ppm ODA. The color comparator increments are: 0, 0.05, 0.10, 0.15, 0.25, 0.50, 1.0 ppm. Technical Support is available for customers who require assistance establishing a FFA correction factor.
For more information, refer to the
K-1001 CHEMets Filming Amine product page

References
- Hater, Wolfgang; Film_Forming Amines: An Innovative Technology for Boiler Water Treatment; The Analyst (Volume 22 Number 4)
- The International Association for the Properties of Water and Steam; Technical Guidance Document: Application of Film Forming Amines in Fossil, Combined Cycle, and Biomass Power Plants; Sept. 2016.
- Odar, Suat; Use of film forming amines (FFA) in nuclear power plants for lay-up and power operation; Advanced Nuclear Technology International, Sweden, Dec. 2017.
- CHEMetrics Inc.; Version 4, Jun 2013, Film Amine (aliphatic amine) Methyl Orange Method Technical Data Sheet, Midland VA.
- ASTM International, 1980, ASTM D 2327-80, Mono- and Dioctadecylamines in Water, West Conshohocken, PA.
Turbidity is haze or cloudiness in water caused by suspended matter. Turbid water is commonly described as hazy, milky, cloudy or dirty. High levels of turbidity usually indicate low water quality.
A common field test to check for turbidity is to hold a flashlight up to the water sample. If the light makes the sample look cloudy or the beam highlights particles in the water, your sample is turbid. Turbidity is scientifically measured using an instrument called a turbidimeter or nephelometer and is reported in Nephelometric Turbidity Units (NTU).
Turbidity interferes with instrumental colorimetric water analysis by causing discoloration and light scattering in the sample. Sample filtration is commonly employed to reduce turbidity, but this is not always appropriate depending on the testing environment or the analyte being measured.
If filtering samples to remove turbidity is not an option when using CHEMetrics Vacu-vials® kits, then the Sample Zeroing Accessory Pack may be your answer. The Sample Zeroing Accessory Pack allows the operator to zero their instrument with a zero blank from the same source as the water sample. This corrects for the turbidity in the sample allowing for a more accurate measurement of the analyte being tested.
For more information, visit us at https://www.chemetrics.com/szap/. Also, watch a brief demonstration HERE
.
As water-based industries continue to expand and evolve, the need for water analysis grows with them. New regulations and market dynamics shift the testing needs to lower cost, more precise readings, and faster reporting. Companies look to high-tech advancements like smart meters and on-line analyzers for efficiency and accuracy but continue to rely on proven colorimetric test methods to meet their needs.

How Colorimetry Works
Colorimetry is the use of colored compounds to determine the concentration of a target chemical compound. It is one of the earliest and most reliable forms of water analysis and is used to test for a wide array of analytes. The target analyte causes the sample solution to change color proportionally to its concentration in the solution, and that change in color can be measured visually or instrumentally.
Traditionally, laboratories stocked the individual chemical components and analysts prepared reagents in bulk from scratch. Often excess expired reagent had to be disposed. Test procedures involved making multiple volume and mass measurements requiring training to master these techniques.

The Benefits of Colorimetric Water Analysis Test Kits
Test kits offer advantages to the analyst in that they save time, are appropriate for field applications of colorimetric water analysis, and are generally user-friendly. Relatively unskilled staff can be trained to make accurate determinations using premixed liquid or solid reagents delivered in kit packaging.
Colorimetric test kits are among the simplest, quickest and most adaptable means by which water analysis may be performed in a host of environments and conditions.
- Tests often require little to no sample preparation and can be performed in minutes.
- Reagents are labeled with expiration dates.
- Technical support is typically available.
- Test kit quality control is the responsibility of the manufacturer.
- Measurement ranges vary depending on test type and analyte, but most tests can measure below one part per million (ppm), allowing for analysis even at low concentrations.
- Colorimetric testing is adaptable to field or lab work.
- Low-cost visual test kits are portable and perfect for testing in the field.
- Instrumental test kits may be better suited for lab work where more precise results are required.
- Many instruments are small and portable, allowing precise results to be gathered in the field with ease.
When using a visual kit, the color of the test result is compared to a chart or comparator of colors to determine the concentration. For instrumental kits, the sample is analyzed in a spectrophotometer or colorimeter. These instruments measure the light absorbance at a specific wavelength which is then converted to the concentration of the analyte.

Colorimetric Water Test Kit Applications
Colorimetric test kits are available for many analytes essential to industrial operations. For example, petroleum refiners commonly measure ammonia, phenols, iron and sulfide. The wastewater treatment industry monitors ammonia, chlorine and phosphates. The medical and food & beverage industries, meanwhile, test for residual disinfectants (chlorine, hydrogen peroxide, ozone, peracetic acid) to ensure sanitization is being performed correctly.
Industrial water treatment professionals are charged with maintaining water treatment programs that help extend equipment life, maximize energy efficiency, and reduce downtime. They are focused on controlling scale, corrosion and microbiological fouling in boiler and cooling systems. They monitor parameters such as dissolved oxygen, bromine, molybdate and various oxygen scavengers in order to make informed decisions about treatment adjustments.

One common application of colorimetric testing is to spot check systems between reporting periods to document whether processes remain under control. Colorimetric test kits are also used alongside on-line analyzers to verify their performance.
Many colorimetric test methods have been reviewed by the USEPA and accepted for monitoring drinking water and wastewater. Examples include chemical oxygen demand (COD) and chlorine.

Conclusion
Colorimetric test kits continue to be practical tools for analysts. Their low cost, convenience, flexibility, accuracy and efficiency appeal to users in all industries. As testing needs evolve, colorimetric water analysis will continue to be an indispensable mainstay in the range of testing options. Analysts have been relying on CHEMetrics and its full line of visual and instrumental colorimetric water testing products for more than 50 years. Visit www.chemetrics.com to learn more.


Congratulations to researchers at China’s Dalian Polytechnic University and the School of Environmental Science and Engineering at Shandong
University. Utilizing CHEMetrics COD water analysis test kits, they recently determined nanogels may be applied to treat pulp bleaching water in a way that makes current wastewater treatments more efficient and environmentally sustainable.
Thanks to these scientists for their work supported in part by the National Science Foundation of China and the Natural Science Foundation of Liaoning. We’re proud to have played a small part in ushering in this advance.
For more information about the research click HERE.
For more information about CHEMetrics full line of Chemical Oxygen Demand test kits click HERE.
MIDLAND, VA – The PAA residual analysis method is now available as a Standard Method thanks in part to the work of CHEMetrics Research and Development Director Joanne Carpenter.

[video_popup url=”https://www.youtube.com/watch?v=pm8X-RrKwZA”]CLICK HERE to watch our video: “Carpenter & CHEMetrics: Leading the Way Toward Greener Wastewater Treatment Methods”[/video_popup]
In collaboration with colleagues from across the wastewater analysis industry and related regulatory agencies, Carpenter led the Standard Methods Joint Task Group to validate a legacy technique for a new application: measurement of Peracetic Acid in water using the DPD Method. The group’s work led to the method’s publication in Standard Methods for the Examination of Water and Wastewater which covers all aspects of water and wastewater analysis techniques. (Standard Methods is a joint publication of the American Public Health Association, the American Water Works Association, and the Water Environment Federation.)
Carpenter also shared the group’s findings in a case study (“DPD Legacy Method Applied to Peracetic Acid”) presented at the 2019 National Environmental Monitoring Conference in Jacksonville, Florida.
“The push to develop a standard method for the analysis of peracetic acid is part of a larger effort to advance PAA as a disinfectant in wastewater treatment,” says Carpenter. “I’m proud to have worked with distinguished colleagues from across the country to complete this important project.”
A workgroup of PAA stakeholders formed in 2017 representing EPA’s Office of Water/Engineering and Analysis Division (EAD), POTW operations personnel, engineering consulting firms, PAA suppliers, and test kit vendors. They began efforts to establish a new method for the analysis of PAA based on the well-established N-N-diethyl–p-phenylenediamine (DPD) method. Bulk wastewater samples solicited from publicly-owned treatment plants were used to prepare test samples for the validation study while test kit vendors provided materials and equipment. The workgroup assembled seven analysts who, under the direction of the study monitor, prepared and distributed blind test samples.
With the development of this new PAA-specific method, wastewater professionals may now accurately measure outflow residuals in order to remain compliant with federal guidelines. Method 4500-PAA can be found at standardmethods.org. To learn more about CHEMetrics’ Peracetic Acid test kit product line, visit the company’s website or email at [email protected].
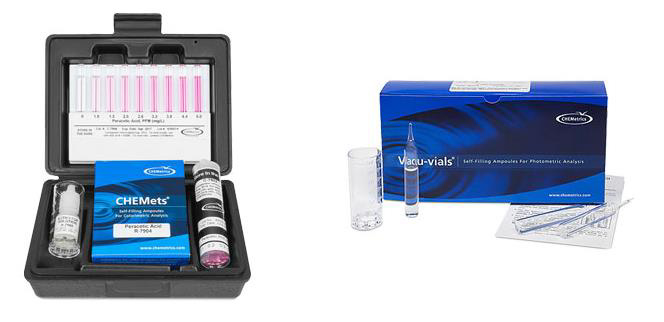
About CHEMetrics
The company’s extensive product line of water and wastewater testing kits utilizes self-filling reagent ampoules to conduct colorimetric water analysis. CHEMetrics® products deliver simplicity, convenience and speed for testing applications in the lab or field. CHEMetrics serves analysts working in aquaculture, boiler/cooling water, chemical processing, drinking water, education, environmental testing, food and beverage, mining, petroleum refining, power generation, pulp and paper, wastewater, and semiconductor manufacturing. Visit www.chemetrics.com to learn more.
CONTACT:
Grant Rampy, Director of Marketing
540.788.9026, x165
[email protected]
Portable DO Test Kits Detect Trace Levels
Application Note
Many power plants use fossil fuels or nuclear power as sources of heat to boil purified water for the production of steam. The pressurized steam drives turbines to produce electricity. The quality of boiler feedwater must be carefully controlled throughout operations to optimize system performance. Proper water quality maintenance maximizes operating efficiency and the lifespan of boiler equipment.
Maintaining Water Quality in High-Pressure Boilers
Dissolved oxygen, or DO, is one of the most important water quality parameters to control in a boiler system. It is the primary corrosive agent of steam-generating systems. Even low concentrations of DO can be highly destructive, causing localized corrosion and pitting of metal system components. Pitting is a concentration of corrosion in a small area of the total metal surface, effectively drilling a hole in the metal. Over time, DO can cause an oxygen tubercle, or scab, to form over the point of original attack. Once the scab forms, the corrosion will continue, even if the system is then properly maintained. The high temperatures and pressures in steam-generating systems accelerate the rate of corrosion. Improper water quality management results in not only inefficiency, but also costly repairs and downtime. Operators of high-pressure boilers try to eliminate DO entirely from feedwater.
Mechanical deaeration is commonly the first and most economical technique employed by plant operators to remove DO. Deaeration equipment heats the feedwater and vents the released gases, including oxygen. Properly maintained deaerators can typically reduce DO levels to as low as 10 ppb (parts per billion).
Plant operators supplement mechanical deaeration with chemical treatment, using oxygen scavengers such as hydrazine, DEHA and carbohydrazide to consume remaining DO. Theoretically, maintaining a high residual of an oxygen scavenger would consume all dissolved oxygen. In reality, competing chemical reactions between oxygen and boiler surfaces are more likely to occur, resulting in significant corrosion within the system.
Consequently, maintaining high scavenger levels may not provide adequate protection. Routine monitoring of DO levels is crucial to confirm sufficient removal of DO within the system.
Common Methods for Monitoring DO
Various kinds of dissolved oxygen monitoring equipment are commercially available. Probes and analyzers with galvanic, polarographic, and optical sensors are often mounted permanently inline. Portable colorimetric test kits that are sensitive enough to measure low levels of dissolved oxygen are also routinely used in boiler applications. Plant engineers consider equipment performance, reliability, measurement frequency, and maintenance costs to determine the most suitable methods for monitoring DO in their systems.
Online analyzers offer continuous DO measurement, but require routine calibration of the sensors. Calibration is typically based on measurements of water-saturated air, which can be accomplished by placing the probe in the air space above the water level in a closed container of water. Calibration frequency depends on vendor guidance, the type of sensor, and the water quality conditions to which the probe is exposed. With the use of either a sensor or an analyzer, it is recommended that plant operators periodically compare online data to results obtained with an alternate test method, ensuring that equivalent results are obtained. This helps to identify calibration drift, sensor corrosion, or other factors that could compromise the validity of online results.
CHEMetrics, LLC is the only manufacturer of a portable test kit for detecting trace levels of dissolved oxygen in boiler applications. CHEMetrics® Test Kits feature the convenience of “snap and read” self-filling ampoules, offering plant operators a rapid, reliable, maintenance-free means of determining ppb levels of DO within one minute. Over the course of nearly 50 years, CHEMetrics has earned a reputation for providing quality DO test kits to this marketplace.
Test kits provide all the components necessary for analysis and do not require calibration by the operator. CHEMetrics ampoules are subjected to a rigorous quality control process where product performance is verified with certified oxygen gas standards, ensuring accurate results. In addition, CHEMetrics is the sole source supplier of the test kits specified in ASTM D5543-15, Standard Test Method for Low-Level Dissolved Oxygen in Water.
CHEMetrics® Test Kits are commonly used as a primary means to measure DO in boiler systems. They are also used as a secondary means to verify online equipment readings or as a backup method when online equipment is out of service.
Sampling and Analysis
Each online analyzer continuously monitors a single location, typically near the discharge side of the boiler feedwater pump. A single CHEMetrics portable test kit, on the other hand, can be moved from one sampling point to another to allow for testing at the deaerator or any potential leakage points throughout the system.
For both online and test kit methods of analysis, it is imperative that the sample water not be exposed to the air. Atmospheric oxygen would contaminate the sample, causing erroneously high results. Therefore, collection of a grab sample is unacceptable for DO analysis.
CHEMetrics test kits are equipped with a special “sampling tube” that is vertically mounted and connected to the sampling port of the boiler system. A continuous flow of sample through the tube prevents contamination from atmospheric oxygen. When the operator submerges the vacuum-sealed ampoule in the flowing sample and snaps the tip, sample is drawn into the ampoule. The highly sensitive colorimetric Rhodazine–D™ reagent reacts instantaneously to produce a pink color.

Conclusion
Dissolved Oxygen must be monitored and controlled to avoid catastrophic failure of steam-generating boiler systems. CHEMetrics’ portable, easy-to-use Dissolved Oxygen Test Kits1 provide plant operators with many advantages when used as a primary monitoring method or alongside online equipment for DO testing.With sensitivity down to 2 ppb, CHEMetrics® ampoules employ an ASTM method for measuring trace levels of DO in boiler applications.

Click HERE for more information on dissolved oxygen and other test kits applicable to industrial water treatment.
1CHEMetrics® Visual Test Kits: K-7511, K-7518, K-7540 and K-7599
CHEMetrics® Hydrogen Peroxide Self-filling Ampoules vs. Test Strips
Aug 2018, Ver 1
Application Note
In the Food and Beverage industry, plant operators on packaging lines routinely monitor sterilization solution residuals in Extended Shelf-Life (ESL) and aseptic packaging applications. The product cartons used to package juice, milk and other dairy products are sprayed with hydrogen peroxide to pre-sterilize them, then heated to remove the hydrogen peroxide. The use of ESL and aseptic processes increases product shelf-life and reduces or eliminates the need for refrigeration or addition of preservatives.
Small amounts of food grade hydrogen peroxide are generally recognized as safe for consumption. However, it is widely understood that appropriate measures should be taken to remove residual peroxide from finished products, as peroxide residual may cause a detrimental effect on nutritional value and loss of quality of the product. The US FDA specifies a limit on hydrogen peroxide residual in packaging of no more than 0.5 parts per million (ppm)1. This peroxide level is often referenced globally as an acceptable residual limit.
Plant operators monitor sterilization residuals by filling representative beverage containers to the normal fill level with distilled water rather than product, then analyzing the distilled water rinsate for hydrogen peroxide. Analytical tools for monitoring residuals include CHEMetrics hydrogen peroxide self-filling ampoules and hydrogen peroxide test strips.
Summary of Evaluation
In this study, comparison testing was performed between CHEMetrics® visual CHEMets® Test Kit, Cat. No. K-5510, CHEMetrics® photometric Vacu-vials® Test Kit, Cat. No. K-5543, and EMD Millipore’s MQuant™ Test Strips (Part No. 110011).
The CHEMetrics® self-filling ampoules and the MQuant™ test strips were evaluated in triplicate at four hydrogen peroxide concentrations: 0, 0.15, 0.25, and 0.50 ppm. Standards were prepared in distilled water. For each replicate, the same aliquot was tested with the test strip and both types of self-filling ampoules.
Color standard increments with K-5510 CHEMets® Kit: 0, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.8 ppm
Scale increments with the MQuant™ Test Strips: 0, 0.5, 2, 5, 10, and 25 ppm
Results for the K-5543 Vacu-vials® Kit were obtained with two CHEMetrics® factory calibrated (direct read) photometers, I-2016 Single Analyte Photometer and V-2000 Multi-Analyte Photometer. Results were obtained in 0.01 ppm increments.
Results
The CHEMetrics® K-5510 visual CHEMets kit was used to easily distinguish concentrations in 0.05 ppm increments in the 0-0.5 ppm range. Picture 1 displays color development at 0.5 ppm. Test results obtained at all check points were exactly equivalent to the actual standard concentration.

Figure 1: CHEMEtrics® C-5501 Hydrogen Peroxide Low Range Color Comparator at 0.5 ppm
Similarly, average results obtained with CHEMetrics® K-5543 photometric Vacu-vials kit were within ± 0.02 ppm of the standard concentration (Table 1).
MQuant™ Test Strips were able to distinguish between 0 ppm and 0.5 ppm hydrogen peroxide. Color intensity with the test strips at 0.25 ppm, though pale, was estimated to fall halfway between the 0 and 0.5 ppm scale increments. At 0.15 ppm, color development with the test strip was barely perceptible. Picture 2 displays the color intensity observed with the MQuant™ Test Strips during testing with all standards. At best, the test strips demonstrated a positive test result at 0.15 ppm.

Figure 2: MQuant™ Test Strips Color Intensity
Conclusion
Precise, accurate and repeatable results are obtained with CHEMetrics® Hydrogen Peroxide Test Kits in the 0-0.5 ppm test range that is critical to routine peroxide residual monitoring in ESL and aseptic packaging applications. The simple snap-and-read technology provides results within 1 minute. The CHEMetrics test kits should be stored in the dark at room temperature.
The MQuant™ Test Strips, although easy to use, are not designed to accurately distinguish results at various sub-ppm levels of peroxide. Due to the broad scale increments and the very pale reaction color below 2 ppm, the test strips should be used only as a presence-absence test in the 0-0.5 ppm range. The test strips require refrigeration as well as humidity and light exposure control.
Table 1: Average Hydrogen Peroxide Test Results CHEMetrics® Hydrogen Peroxide Self-filling Ampoules vs. Test Strips.
|
Peroxide Conc., ppm |
CHEMetrics K-5510, ppm |
CHEMetrics K-5543, ppm |
MQuant™ Test Strips, ppm |
|
0 |
0 | 0.00 | 0 |
|
0.15 |
0.15 | 0.13 |
> 0 |
|
0.25 |
0.25 | 0.24 | 0.25 |
| 0.50 | 0.50 | 0.48 |
0.5 |
1Code of Federal Register, Title 21, Part 178, Section 178.1005(d)
Vacu-vials® and CHEMets® are registered trademarks of CHEMetrics, LLC
MQuant™ is a trademark of EMD Millipore.
© 2018 CHEMetrics, LLC

